Media Summary: As a healthcare provider, navigating the "massive rule book" that governs prescribing is absolutely critical for keeping both SUBSCRIBE to to see more videos. Details and supporting materials: ... Frier Levitt attorneys present "Understanding
Your Fda Compliance Checklist Off Label Compounding - Detailed Analysis & Overview
As a healthcare provider, navigating the "massive rule book" that governs prescribing is absolutely critical for keeping both SUBSCRIBE to to see more videos. Details and supporting materials: ... Frier Levitt attorneys present "Understanding Nick Capman talks with Jesse Gillikan, President of cGMP Validation, about the challenges Viewers will hear an overview of Environmental Monitoring for Salvatore Dileo, RPh, Director of Pharmacy, Westmed Medical Group in New York, discusses the differences between ...
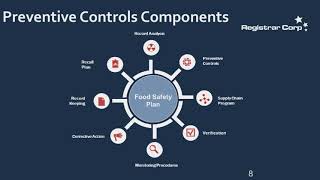
Stay ahead of the game with this quick dive into Drug Registration and Listing Staff Lysette Deshields and Soo Jin Park provide a walk-through of the creation of a 503B product ... To learn more about FDAs Inspection Process For Under the Food Safety Modernization Act (“FSMA”), certain food facilities registered with the U.S.